When it comes to Equipment Qualification, Philip says STERIS is “in a unique position” due to its “multi-discipline engineering team” which makes up mechanical, biomedical, electronic and quality engineers. “It also introduces testing at various stages of the Product Lifecycle and ensures their accuracy while creating a solid foundation for traceable evidence practices throughout the lifecycle.”

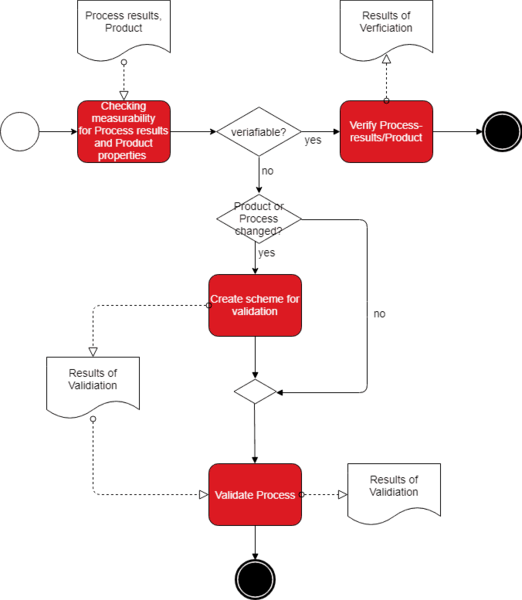
Test Method Validation confirms that a test method can consistently provide valid and compliant results,” explained Philip. “Under any of these circumstances, employing Test Method Validation would be an advisable course of action.
Iso 13485 test method validation verification#
STERIS’s services include Test Method Validation (TMV) and verification along with equipment qualification. Radiation Request Form Submittal Guidelinesįailure to maintain consistent and traceable test processes at all stages of the product lifecycle is a recipe for disaster, according to the test engineers at STERIS.Īs a test laboratory itself, Galway-based STERIS knows only too well how important it is that all test processes utilized by a company deliver consistent, accurate and traceable test results.Sterile Barrier Integrity and Seal Strength.Dangerous Goods (UN DG) Package Testing.Method Development and Validation Services.Radiation Sterilization Validation Services.Sustainable EO ® Sterilization Services.
